| Structure | Name/CAS No. | Articles |
|---|---|---|
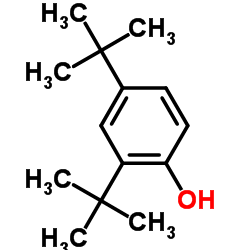 |
2,4-Di-t-butylphenol
CAS:96-76-4 |
|
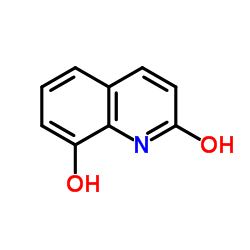 |
2,8-Dihydroxyquinoline
CAS:15450-76-7 |