Location of naphthol yellow-S binding site on bovine serum albumin.
M Iwatsuru, H Nishigori, K Maruyama
Index: J. Pharmacobiodyn. 4(11) , 851-9, (1981)
Full Text: HTML
Abstract
The characteristics of the binding site in the first binding class of naphthol yellow-S (NY-S) on bovine serum albumin (BSA) were studied. The binding of NY-S to BSA at an equimolar ratio of each material resulted in a marked quenching of intrinsic fluorescence of BSA and a decrease in the binding capacity of 1-anilinonaphthalene-8-sulfonate to BSA. The binding of NY-S to BSA was diminished by the chemical modification of tryptophan residue in the BSA molecule with 2-hydroxy-5-nitrobenzyl bromide and o-nitrophenylsulfenyl chloride. The higher modifications rate of tryptophan residue decreased the binding constant of NY-S to BSA. These results suggest that the first binding site of NY-S to BSA is located in a hydrophobic area including tryptophan which is position 134 on the amino acid sequence of BSA. Studies on BSA modified with diethylpyrocarbonate demonstrated that a histidine residue also may participate in the binding of NY-S to BSA.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
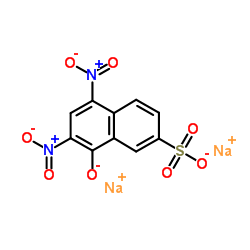 |
Citronin A
CAS:846-70-8 |
C10H4N2Na2O8S |
|
Determination of the fraction of G0 cells in cytologic sampl...
1983-03-01 [Anal. Quant. Cytol. 5(1) , 1-4, (1983)] |
|
"In situ"--measurements of protein contents in the brush bor...
1981-01-01 [Histochemistry 72(3) , 467-79, (1981)] |
|
Quantitative cytochemical differences between young and old ...
1985-12-01 [Chest 88(6) , 864-9, (1985)] |
|
Quantification of nuclear non-histone proteins by Feulgen--N...
1981-09-01 [Histochem. J. 13(5) , 717-22, (1981)] |
|
Automated selection of the most epithelium-rich areas in gyn...
1991-12-01 [Anal. Quant. Cytol. Histol. 13(6) , 395-402, (1991)] |