Stereochemical surprises in the Lewis acid-mediated allylation of isatins.
Devendra J Vyas, Roland Fröhlich, Martin Oestreich
Index: J. Org. Chem. 75(19) , 6720-3, (2010)
Full Text: HTML
Abstract
The BF(3)·OEt(2)-mediated allylation of isatin with an α-chiral allylic stannane is diastereo- and enantioselective. Conversely, allylation of any substituted isatin employing the identical protocol is not diastereoselective at all and only enantioselective for the major diastereomer having syn relative configuration. The anti isomer is, however, formed in almost racemic form. Both absolute and relative configurations are unambiguously secured by X-ray analysis of major isomers, and the stereochemical assignment of the other 3-substituted 3-hydroxy oxindoles is based on similar NMR spectroscopic characteristics. The remarkable observations are rationalized by an acyclic transition state model.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
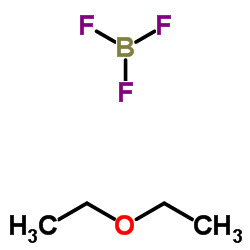 |
Boron trifluoride etherate
CAS:109-63-7 |
C4H10BF3O |
|
Carbohydrate-Specific Uptake of Fucosylated Polymeric Micell...
2015-07-13 [Biomacromolecules 16 , 1948-57, (2015)] |
|
Carbohydrate conjugation through microwave-assisted function...
2015-03-20 [Carbohydr. Res. 405 , 33-8, (2015)] |
|
Pegylation effect of chitosan based polyplex on DNA transfec...
2015-04-20 [Carbohydr. Polym. 120 , 7-14, (2015)] |
|
Rapid conversion of spirostans into furostan skeletons at ro...
2012-01-01 [Steroids 77(1-2) , 59-66, (2012)] |
|
Tailoring Oxygen Sensitivity with Halide Substitution in Dif...
2015-10-28 [ACS Appl. Mater. Interfaces 7 , 23633-43, (2015)] |