Synthesis of a new amphiphilic glycodendrimer with antiviral functionality.
Shuqin Han, Taisei Kanamoto, Hideki Nakashima, Takashi Yoshida
Index: Carbohydr. Polym. 90(2) , 1061-8, (2012)
Full Text: HTML
Abstract
A new third generation amphiphilic glycodendrimer was synthesized from a stearylamide lysine dendrimer by condensation of the oligosaccharide moiety. By stepwise condensation and deprotection of di-boc lysine from a core of stearyl amide lysine, a third-generation stearylamide lysine dendrimer was constructed. Acetyl cellobiose and glucose units with the carboxylic acid at the end of alkyl chain attached to the reducing end of the sugar moiety was condensed with surface amino groups of the third generation lysine dendrimer, respectively, to give a new stearylamide acetylcellobiose and acetylglucose lysine dendrimers. The structural analysis was carried out using NMR, IR, and matrix-associated laser desorption/ionization time-of-flight (MALDI TOF) mass spectroscopies. After deacetylation to recover hydroxyl groups and subsequent sulfation, the third-generation sulfated cellobiose stearylamide lysine dendrimer was preliminarily found to have high anti-HIV activity at a 50% effective concentration (EC(50)) as low as 6.4 μg/ml and low cytotoxicity at a 50% cytotoxic concentration (CC(50)) as high as 1000 μg/ml, indicating that the dendrimer gave the enhancement of the functionality of oligosaccharides with low molecular weights. The glycodendrimer with a hydrophobic stearyl chain is immobilized on hydrophobic surfaces by hydrophobic interaction and is expected to provide a new biomedical material with the surface functionality of hydrophilic sulfated oligosaccharides.Copyright © 2012 Elsevier Ltd. All rights reserved.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
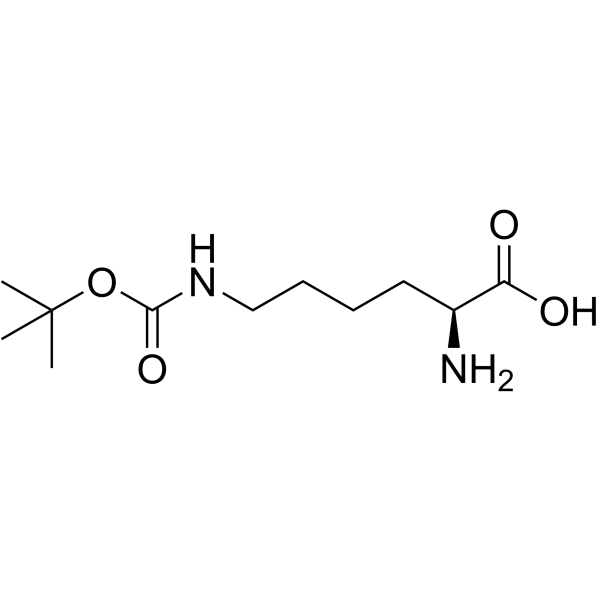 |
Ne-Boc-L-lysine
CAS:2418-95-3 |
C11H22N2O4 |
|
Adding l-lysine derivatives to the genetic code of mammalian...
2008-07-11 [Biochem. Biophys. Res. Commun. 371(4) , 818-22, (2008)] |
|
Evidence that malondialdehyde-derived aminoenimine is not a ...
2001-05-01 [Chem. Res. Toxicol. 14(5) , 473-5, (2001)] |
|
Nanoassembly of surfactants with interfacial drug-interactiv...
2013-01-07 [Mol. Pharm. 10(1) , 187-98, (2013)] |
|
Formation of glyoxal, methylglyoxal and 3-deoxyglucosone in ...
1999-11-15 [Biochem. J. 344 Pt 1 , 109-16, (1999)] |
|
Chain-length dependence for secondary structure formation of...
1984-01-01 [Int. J. Pept. Protein Res. 23(1) , 47-54, (1984)] |