Carbofuran metabolism and toxicity in the rat.
P W Ferguson, M S Dey, S A Jewell, R I Krieger
Index: Fundam. Appl. Toxicol. 4(1) , 14-21, (1984)
Full Text: HTML
Abstract
The influence of carbofuran metabolism on acetylcholinesterase inhibition has been defined after low dose (50 micrograms/kg, iv and oral) [carbonyl-14C]carbofuran exposures to male Sprague-Dawley rats. Red blood cell acetylcholinesterase (RBC AchE) inhibition (83% at 2 min, 37% at 15 min for iv and oral, respectively, with recovery by 3 hr), was correlated with carbofuran plasma concentrations (r = 0.97). Eight-hour sample collection indicated that ultimate carbofuran fate (41-47% 14CO2, 14-15% urine, less than 1% feces, and 30-31% carcass) was independent of exposure route. Carbofuran absorption (peak plasma levels less than 7 min), distribution, and elimination (t1/2 = 29 +/- 5 min) occurred rapidly. 3-Hydroxycarbofuran, a significant oxidative metabolite of carbofuran with anticholinesterase activity, was rapidly formed and subject to enterohepatic circulation (plasma t1/2 = 64 +/- 5 min). Results indicated that rapid RBC AchE recovery closely paralleled carbofuran metabolism and the primary in vivo disposition of 3-hydroxycarbofuran was metabolic conjugation.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
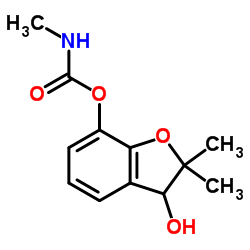 |
3-Hydroxy Carbofuran
CAS:16655-82-6 |
C12H15NO4 |
|
Determination of pesticides in soy-based infant formula usin...
2006-01-01 [J. AOAC Int. 89(1) , 214-24, (2006)] |
|
Metabolism of carbofuran by Aspergillus niger and Fusarium g...
1998-05-01 [J. Environ. Sci. Health B 33(3) , 253-66, (1998)] |
|
Surprising hair analysis results following acute carbofuran ...
2011-10-10 [Forensic Sci. Int. 212(1-3) , e10-4, (2011)] |
|
Residues of carbofuran and its two carbamate metabolites in ...
1983-01-01 [J. Environ. Sci. Health B 18(2) , 301-4, (1983)] |
|
[Determination of furadan and its metabolites in biomedia by...
1982-07-01 [Gig. Sanit. (7) , 45-6, (1982)] |