Determination of midazolam and 1-hydroxymidazolam from plasma by gas chromatography coupled to methane negative chemical ionization mass spectrometry after sublingual administration of midazolam.
Ruut Kaartama, Pekka Jarho, Jouko Savolainen, Hannu Kokki, Marko Lehtonen
Index: J. Chromatogr. B. Analyt. Technol. Biomed. Life Sci. 879(19) , 1668-76, (2011)
Full Text: HTML
Abstract
A sensitive and selective gas chromatographic mass spectrometric method for the determination of midazolam and its biologically active metabolite, 1-hydroxymidazolam, in rabbit plasma has been developed and validated. Sample preparation includes mixed-mode solid-phase extraction and derivatization with silylating reagents. Midazolam-d4 was used as an internal standard for the determination of parent drug and its active metabolite. The instrumentation consisted of a capillary column gas chromatography and a single quadrupole mass spectrometer with a negative chemical ionization. The method was found to be valid in terms of selectivity, linearity, precision, accuracy, and recovery over the concentration range of 2-200 ng/ml and 1-100 ng/ml for midazolam and 1-hydroxymidazolam, respectively. For both analytes, the lower limit of quantification was 2 ng/ml. Midazolam was stable in stock solutions stored three months at -20°C and in human plasma stored for three months at -80°C. In addition, no degradation of midazolam was found after three freeze-thaw cycles, in short-term stability at room temperature for 24h, or in post-preparative stability in the autosampler. The validity of the method was further tested by performing a pharmacokinetic study of sublingual administration of midazolam in rabbits. The method will be used in studies related to a formulation development of novel midazolam formulations for use in paediatric anaesthesia.Copyright © 2011 Elsevier B.V. All rights reserved.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
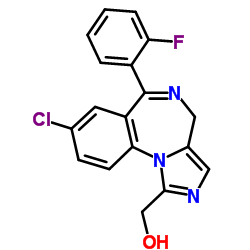 |
1'-Hydroxymidazolam
CAS:59468-90-5 |
C18H13ClFN3O |
|
A comparison of the metabolism of midazolam in C57BL/6J and ...
2014-12-15 [Biochem. Pharmacol. 92(4) , 701-11, (2014)] |
|
A highly sensitive liquid chromatography tandem mass spectro...
2011-10-01 [Biomed. Chromatogr. 25(10) , 1091-8, (2011)] |
|
Evidence of CYP3A allosterism in vivo: analysis of interacti...
2012-03-01 [Clin. Pharmacol. Ther. 91(3) , 442-9, (2012)] |
|
Metabolism of 1'- and 4-hydroxymidazolam by glucuronide conj...
2010-11-01 [Drug Metab. Dispos. 38(11) , 2007-13, (2010)] |
|
The structural basis for homotropic and heterotropic coopera...
2011-12-20 [Biochemistry 50(50) , 10804-18, (2011)] |