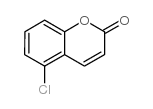
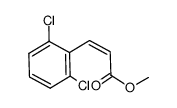
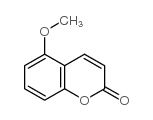
|
~% |
|
~% |
|
~% |
|
~82% |
|
~% |