Classical Example of Total Kinetic and Thermodynamic Control: The Diels–Alder Reaction between DMAD and Bis-furyl Dienes
KseniyaK. Borisova, Elizaveta A. Kvyatkovskaya, Eugeniya V. Nikitina, Rinat R. Aysin, Roman A. Novikov, Fedor I. Zubkov
Index: 10.1021/acs.joc.8b00336
Full Text: HTML
Abstract
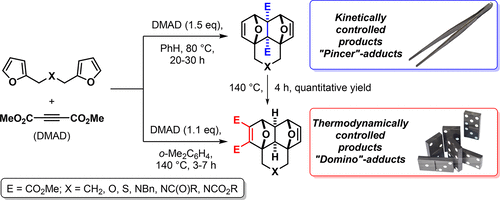
A rare example of chemospecificity in the tandem Diels–Alder reaction of activated alkynes and bis-dienes has been revealed. The reaction between bis-furyl dienes and DMAD occurs at 25–80 °C and leads to kinetically controlled “pincer” adducts, 4a,8a-disubstituted 1,4:5,8-diepoxynaphthalenes. On the contrary, only thermodynamically controlled “domino” adducts (2,3-disubstituted 1,4:5,8-diepoxynaphthalenes) are formed in the same reaction at 140 °C. The “pincer” adducts can be transformed to the “domino” adducts at heating. The rate constants for reactions of both types were calculated using dynamic 1H NMR spectroscopy.
|
Synthesis and Unique Optical Properties of Selenophenyl BODI...
2018-04-19 [10.1021/acs.joc.8b00782] |
|
Synthesis of 3,5-Disubstituted BODIPYs Bearing N-Containing ...
2018-04-17 [10.1021/acs.joc.8b00087] |
|
Steric Hindrance Underestimated: It is a Long, Long Way to T...
2018-04-17 [10.1021/acs.joc.8b00496] |
|
Stereoselective Sequential [4+2]/[2+2] Cycloadditions Involv...
2018-04-16 [10.1021/acs.joc.8b00346] |
|
Pd-Induced Double C–H Bond Activation in Annulative Synthese...
2018-04-16 [10.1021/acs.joc.8b00630] |