The Journal of Organic Chemistry
2018-04-04
Metal-Free Iodine-Catalyzed Oxidation of Ynamides and Diaryl Acetylenes into 1,2-Diketo Compounds
SeungWoo Kim, Tae-Woong Um, Seunghoon Shin
Index: 10.1021/acs.joc.8b00484
Full Text: HTML
Abstract
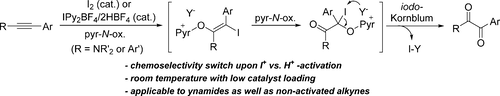
Metal-free oxidation of ynamides is described, employing pyridine-N-oxides as oxidants under molecular iodine catalysis. In stark contrast to Brønsted acid catalysis, iodophilic activation of ynamides diverts the reaction manifold into a dioxygenation pathway. This oxidation is very rapid at room temperature with only 2.5 mol % I2. Furthermore, this protocol could be extended to nonactivated alkynes, such as diarylacetylenes, to deliver various benzil derivatives.
Latest Articles:
More...
|
Synthesis and Unique Optical Properties of Selenophenyl BODI...
2018-04-19 [10.1021/acs.joc.8b00782] |
|
Synthesis of 3,5-Disubstituted BODIPYs Bearing N-Containing ...
2018-04-17 [10.1021/acs.joc.8b00087] |
|
Steric Hindrance Underestimated: It is a Long, Long Way to T...
2018-04-17 [10.1021/acs.joc.8b00496] |
|
Stereoselective Sequential [4+2]/[2+2] Cycloadditions Involv...
2018-04-16 [10.1021/acs.joc.8b00346] |
|
Pd-Induced Double C–H Bond Activation in Annulative Synthese...
2018-04-16 [10.1021/acs.joc.8b00630] |