Efaroxan hydrochloride
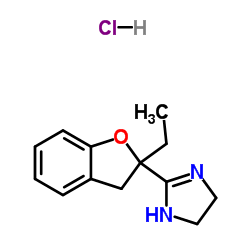
Efaroxan hydrochloride structure
|
Common Name | Efaroxan hydrochloride | ||
|---|---|---|---|---|
| CAS Number | 89197-00-2 | Molecular Weight | 252.740 | |
| Density | N/A | Boiling Point | 387ºC at 760 mmHg | |
| Molecular Formula | C13H17ClN2O | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 187.9ºC | |
| Symbol |

GHS06 |
Signal Word | Danger | |
Use of Efaroxan hydrochlorideEfaroxan hydrochloride is a potent and selective α2-adrenoceptor antagonist, antidiabetic activity. Efaroxan hydrochloride is a selective I1-Imidazoline receptor antagonist and can be used for the research of cardiovascular disease[1][2][3]. |
| Name | 2-(2-ethyl-3H-1-benzofuran-2-yl)-4,5-dihydro-1H-imidazole,hydrochloride |
|---|---|
| Synonym | More Synonyms |
| Description | Efaroxan hydrochloride is a potent and selective α2-adrenoceptor antagonist, antidiabetic activity. Efaroxan hydrochloride is a selective I1-Imidazoline receptor antagonist and can be used for the research of cardiovascular disease[1][2][3]. |
|---|---|
| Related Catalog | |
| Target |
α2-adrenoceptor, I1-Imidazoline receptor[1][2] |
| In Vitro | Efaroxan hydrochloride binds to I1-imidazoline and α2-adrenergic receptors in bovine rostral ventrolateral medulla membranes, with Kis of 0.15 and 5.6 nM, respectively[1] |
| In Vivo | Efaroxan hydrochloride increases plasma insulin levels in both conscious fed[3]. Animal Model: Male Sprague-Dawley rats (weight range 250-300g)[3] Dosage: 1 mg/kg, 5 mg/kg Administration: Oral administration Result: Significant increase in plasma insulin levels of starved rats 15 and 30 min after treatment. |
| References |
| Boiling Point | 387ºC at 760 mmHg |
|---|---|
| Molecular Formula | C13H17ClN2O |
| Molecular Weight | 252.740 |
| Flash Point | 187.9ºC |
| Exact Mass | 252.102936 |
| PSA | 33.62000 |
| LogP | 2.33840 |
| HS Code | 2934999090 |
|---|---|
| Summary | 2934999090. other heterocyclic compounds. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
In vivo evaluation of a new ¹⁸F-labeled PET ligand, [¹⁸F]FEBU, for the imaging of I₂-imidazoline receptors.
Nucl. Med. Biol. 42(4) , 406-12, (2015) The functions of I₂-imidazoline receptors (I₂Rs) are unknown, but evidence exists for their involvement in various neuropsychiatric disorders. Although a few positron emission tomography (PET) I₂R lig... |
|
|
Repeated exposure to MDMA triggers long-term plasticity of noradrenergic and serotonergic neurons.
Mol. Psychiatry 19(7) , 823-33, (2014) 3,4-Methylenedioxymethamphetamine (MDMA or 'ecstasy') is a psychostimulant drug, widely used recreationally among young people in Europe and North America. Although its neurotoxicity has been extensiv... |
|
|
Evidence for imidazoline receptors involvement in the agmatine antidepressant-like effect in the forced swimming test.
Eur. J. Pharmacol. 565(1-3) , 125-31, (2007) This study investigated the involvement of the imidazoline receptors in the antidepressant-like effect of agmatine in the forced swimming test. The antidepressant-like effects of agmatine (10 mg/kg, i... |
| Calmidazolium chloride |
| (±)-efaroxan hydrochloride |
| 2-(2-Ethyl-2,3-dihydro-1-benzofuran-2-yl)-4,5-dihydro-1H-imidazole hydrochloride (1:1) |
| Efaroxan HCl |
| 1H-Imidazole, 2-(2-ethyl-2,3-dihydro-2-benzofuranyl)-4,5-dihydro-, hydrochloride (1:1) |
| Efaroxan hydrochloride |