Ethyl gallate
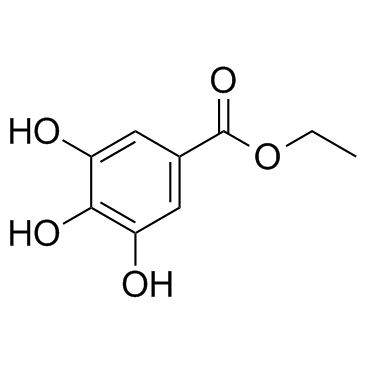
Ethyl gallate structure
|
Common Name | Ethyl gallate | ||
|---|---|---|---|---|
| CAS Number | 831-61-8 | Molecular Weight | 198.173 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 447.3±40.0 °C at 760 mmHg | |
| Molecular Formula | C9H10O5 | Melting Point | 149-153 °C | |
| MSDS | Chinese USA | Flash Point | 185.0±20.8 °C | |
Use of Ethyl gallateEthyl gallate is a nonflavonoid phenolic compound and also a scavenger of hydrogen peroxide. |
| Name | ethyl gallate |
|---|---|
| Synonym | More Synonyms |
| Description | Ethyl gallate is a nonflavonoid phenolic compound and also a scavenger of hydrogen peroxide. |
|---|---|
| Related Catalog | |
| In Vitro | Ethyl gallate is a nonflavonoid phenolic compound and also a scavenger of hydrogen peroxide. After treatment for 24 h or 48 h with Ethyl gallate, HL-60 cells show changes in morphology, including shrinkage of the cell membrane and the development of apoptotic bodies. Consistent with these effects, the viability of Ethyl gallate-treated cells decreases in a time- and dose-dependent manner, demonstrating that Ethyl gallate has a cytotoxic effect on HL-60 cells. Ethyl gallate treatment increases the proportion of cells in subG1 phase in a concentration- and time-dependent manner. Treatment of cells for 24 h or 48 h with 50 μM or 75 μM Ethyl gallate increases the percentage of cells in the subG1 phase from a baseline of 2.9% to 26.5% or 52.6%, respectively. It is found that Ethyl gallate treatment of HL-60 cells decreases the expression of Bcl-2 at 75 μM Ethyl gallate, and increases Bax and truncated Bid (tBid) expression at 24 h[1]. |
| In Vivo | No significant difference in the serum total protein, albumin, globulin and glucose is found between the rats fed with A. nilotica (L.) leaf extract on ethyl gallate equivalent basis and those fed with Ethyl gallate alone. Significant differences in total bilirubin level, however, exist between the rats that receive A. nilotica (L.) leaf extract, 500 mg/kg body weight (ethyl gallate equivalent of 10 mg/kg, 0.34±0.01 mg/dL) and those receiving 10 mg/kg body weight of Ethyl gallate (0.26±0.01 mg/dL). Significant difference is found for ALT between groups fed with 500 and 1000 mg/kg body weight of A. nilotica (L.) leaf extract (26.52±1.23 and 30.05±1.38 U/L) and 10 and 20 mg/kg of Ethyl gallate (20.50±0.94 and 24.67±1.13 U/L)[2]. |
| Kinase Assay | The expression of apoptosis-related proteins (caspases-8, -9, -3; AIF; Endo G; Bid; Bax; and Bcl-2) in HL-60 cells is determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) of lysates followed by western blotting. For this, HL-60 cells (1.5×106) are treated with 50 μM or 75 μM Ethyl gallate for 6 h, 12 h, or 24 h. Total cell lysates are obtained by resuspending cells in ice-cold radioimmunoprecipitation assay (RIPA) buffer for 30 min followed by centrifugation. Protein concentration is determined using a NanoDrop spectrophotometer. Aliquots of lysates (100 μg protein equivalents) are resolved by 12% SDS-PAGE and transferred onto nitrocellulose membranes[1]. |
| Cell Assay | HL-60 cells (1×106) are treated with 50 μM or 75 μM Ethyl gallate for 24 h or 48 h at 37°C. Cells are then harvested by centrifugation and fixed in 70% ethanol at 4°C for 24 h. Fixed cells are resuspended in PBS containing 40 μg/mL Propidium iodide (PI), 100 μg/mL RNase A, and 0.1% Triton X-100 and incubated in the dark for 30 min at room temperature. Cell cycle distribution is analyzed by flow cytometry on a FACSCalibur. To investigate apoptotic cells, HL-60 cells (1×106) incubated with different concentration of 50 μM, 75 μM and 100 μM Ethyl gallate for 24 h or 48 h at 37°C, and then DAPI staining is conducted. The cells are photographed using a fluorescence microscopy[1]. |
| Animal Admin | Forty eight female albino Wistar rats of six to eight weeks old are used and divided into eight groups based on their body weights. Group 1 rats serve as control receiving 1.0 mL of the vehicle (0.1% ethanol); Group 2 rats receive A. nilotica (L.) leaf extract (250 mg/kg body weight); Group 3 rats receive A. nilotica (L.) leaf extract (500 mg/kg body weight); Group 4 rats receive A. nilotica (L.) leaf extract (1000 mg/kg body weight); Group 5 rats receive A. nilotica (L.) leaf extract (2000 mg/kg body weight); Group 6 rats receive Ethyl gallate (5 mg/kg body weight); Group 7 rats receive Ethyl gallate (10 mg/kg body weight); Group 8 rats receive Ethyl gallate (20 mg/kg body weight). Body weights are recorded on 0th and 14th day for each group and all rats are decapitated after an overnight fast[2]. |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 447.3±40.0 °C at 760 mmHg |
| Melting Point | 149-153 °C |
| Molecular Formula | C9H10O5 |
| Molecular Weight | 198.173 |
| Flash Point | 185.0±20.8 °C |
| Exact Mass | 198.052826 |
| PSA | 86.99000 |
| LogP | 2.07 |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.611 |
| Water Solubility | soluble |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
|
~93% 
Ethyl gallate CAS#:831-61-8 |
| Literature: Takaoka, Shigeki; Takaoka, Noriko; Minoshima, Yuka; Huang, Jian-Mei; Kubo, Miwa; Harada, Kenichi; Hioki, Hideaki; Fukuyama, Yoshiyasu Tetrahedron, 2009 , vol. 65, # 40 p. 8354 - 8361 |
|
~90% 
Ethyl gallate CAS#:831-61-8 |
| Literature: Dodo, Kosuke; Minato, Taro; Noguchi-Yachide, Tomomi; Suganuma, Masami; Hashimoto, Yuichi Bioorganic and Medicinal Chemistry, 2008 , vol. 16, # 17 p. 7975 - 7982 |
|
~82% 
Ethyl gallate CAS#:831-61-8 |
| Literature: Ambika; Singh, Pradeep Pratap; Chauhan Synthetic Communications, 2008 , vol. 38, # 6 p. 928 - 936 |
|
~% 
Ethyl gallate CAS#:831-61-8 |
| Literature: Synthetic Communications, , vol. 29, # 8 p. 1405 - 1408 |
| Precursor 5 | |
|---|---|
| DownStream 10 | |
| HS Code | 2918290000 |
|---|---|
| Summary | HS: 2918290000 other carboxylic acids with phenol function but without other oxygen function, their anhydrides, halides, peroxides, peroxyacids and their derivatives Tax rebate rate:9.0% Supervision conditions:AB(certificate of inspection for goods inward,certificate of inspection for goods outward) VAT:17.0% MFN tariff:6.5% General tariff:30.0% |
|
Antimicrobial activity of natural products from the flora of Northern Ontario, Canada.
Pharm. Biol. 53(6) , 800-6, (2015) The number of multidrug resistant (MDR) microorganisms is increasing and the antimicrobial resistance expressed by these pathogens is generating a rising global health crisis. In fact, there are only ... |
|
|
Investigating the potential of under-utilised plants from the Asteraceae family as a source of natural antimicrobial and antioxidant extracts.
Food Chem. 161 , 79-86, (2014) Antimicrobial properties of ethanol and water extracts from eight Asteraceae species were investigated against three Gram positive (Staphylococcus aureus, MRSA and Bacillus cereus) and two Gram negati... |
|
|
The tomato sauce making process affects the bioaccessibility and bioavailability of tomato phenolics: a pharmacokinetic study.
Food Chem. 173 , 864-72, (2014) Tomato sauce is the most commonly consumed processed tomato product worldwide, but very little is known about how the manufacturing process may affect the phenolic composition and bioavailability afte... |
| N-ETHYLGALLATE |
| nipa48 |
| MFCD00016430 |
| nipagallina |
| ethyl 3,4,5-trihydroxybenzoate |
| Nipa No. 48 |
| 3,4,5-Trihydroxybenzoic acid ethyl ester |
| 3,4,5-trihydroxyethylbenzoate |
| Progallin A |
| Gallic acid ethyl ester |
| Phyllemblin |
| Gallic acid ethyl |
| EINECS 212-608-5 |
| UNII-235I6UDD3L |
| Ethyl gallate |
| Benzoic acid, 3,4,5-trihydroxy-, ethyl ester |
| ethylgallate |
![2,6-ditert-butyl-4-[2-(3,5-ditert-butyl-4-hydroxyphenyl)sulfanylpropan-2-ylsulfanyl]phenol structure](https://www.chemsrc.com/caspic/174/100079-21-8.png)
 CAS#:82-12-2
CAS#:82-12-2 CAS#:87-66-1
CAS#:87-66-1 CAS#:5782-85-4
CAS#:5782-85-4 CAS#:476-66-4
CAS#:476-66-4 CAS#:342886-38-8
CAS#:342886-38-8 CAS#:31374-71-7
CAS#:31374-71-7 CAS#:6178-44-5
CAS#:6178-44-5 CAS#:138433-01-9
CAS#:138433-01-9 CAS#:126229-90-1
CAS#:126229-90-1
