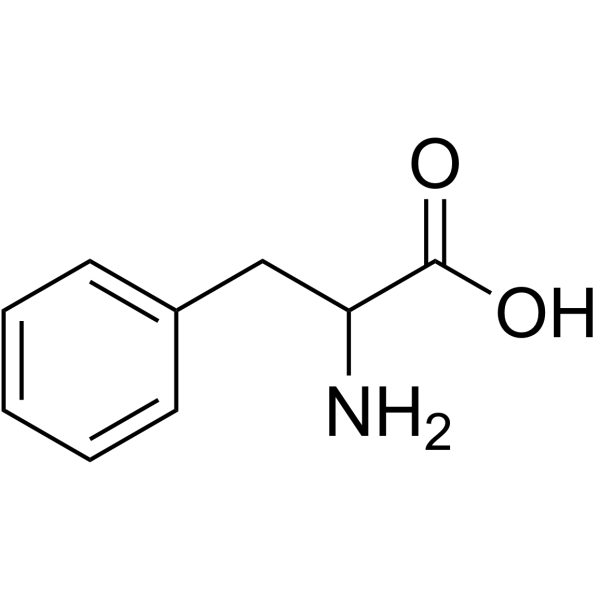
H-Gly-DL-Phe-OH
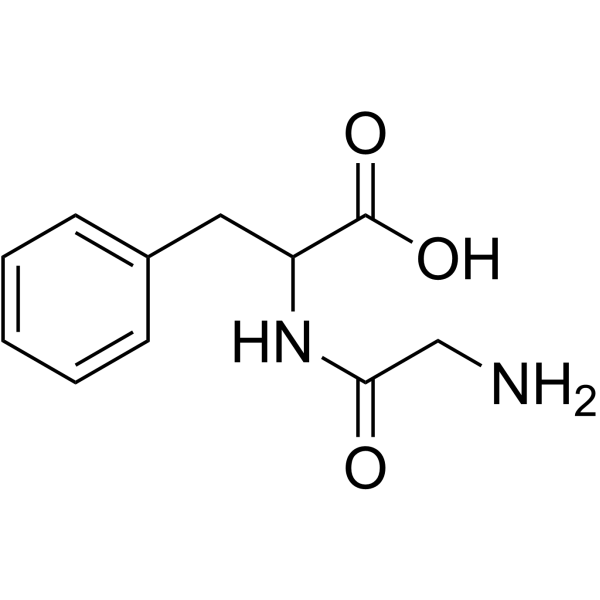
H-Gly-DL-Phe-OH structure
|
Common Name | H-Gly-DL-Phe-OH | ||
|---|---|---|---|---|
| CAS Number | 721-66-4 | Molecular Weight | 222.24000 | |
| Density | 1.259 g/cm3 | Boiling Point | 492.2ºC at 760 mmHg | |
| Molecular Formula | C11H14N2O3 | Melting Point | 273-275℃ (Decomposition) | |
| MSDS | N/A | Flash Point | 251.5ºC | |
Use of H-Gly-DL-Phe-OHGlycyl-DL-phenylalanine is a phenylalanine derivative[1]. |
| Name | Glycyl-DL-phenylalanine |
|---|---|
| Synonym | More Synonyms |
| Description | Glycyl-DL-phenylalanine is a phenylalanine derivative[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Amino acids and amino acid derivatives have been commercially used as ergogenic supplements. They influence the secretion of anabolic hormones, supply of fuel during exercise, mental performance during stress related tasks and prevent exercise induced muscle damage. They are recognized to be beneficial as ergogenic dietary substances[1]. |
| References |
| Density | 1.259 g/cm3 |
|---|---|
| Boiling Point | 492.2ºC at 760 mmHg |
| Melting Point | 273-275℃ (Decomposition) |
| Molecular Formula | C11H14N2O3 |
| Molecular Weight | 222.24000 |
| Flash Point | 251.5ºC |
| Exact Mass | 222.10000 |
| PSA | 92.42000 |
| LogP | 0.84840 |
| Storage condition | −20°C |
| RIDADR | NONH for all modes of transport |
|---|---|
| WGK Germany | 3 |
| HS Code | 2924299090 |
|
~% 
H-Gly-DL-Phe-OH CAS#:721-66-4 |
| Literature: Journal of the American Chemical Society, , vol. 75, p. 5323,5326 |
|
~% 
H-Gly-DL-Phe-OH CAS#:721-66-4 |
| Literature: Chemische Berichte, , vol. 37, p. 3307 |
|
~% 
H-Gly-DL-Phe-OH CAS#:721-66-4 |
| Literature: Chemische Berichte, , vol. 98, p. 3462 - 3469 |
|
~% 
H-Gly-DL-Phe-OH CAS#:721-66-4 |
| Literature: Justus Liebigs Annalen der Chemie, , vol. 498, p. 50,55 |
|
~% 
H-Gly-DL-Phe-OH CAS#:721-66-4 |
| Literature: Justus Liebigs Annalen der Chemie, , vol. 712, p. 208 - 213 |
|
~% 
H-Gly-DL-Phe-OH CAS#:721-66-4 |
| Literature: Chemische Berichte, , vol. 37, p. 3307 |
|
~% 
H-Gly-DL-Phe-OH CAS#:721-66-4 |
| Literature: Journal of the Chemical Society, , p. 642,644 |
|
~% 
H-Gly-DL-Phe-OH CAS#:721-66-4 |
| Literature: Chemische Berichte, , vol. 101, p. 1257 - 1262 |
| HS Code | 2924299090 |
|---|---|
| Summary | 2924299090. other cyclic amides (including cyclic carbamates) and their derivatives; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Riboflavin-binding protein is a novel bitter inhibitor.
Chem. Senses 33(1) , 57-63, (2008) Riboflavin-binding protein (RBP) from chicken egg, which was recently reported to be a selective sweet inhibitor for protein sweeteners, was also found to be a bitter inhibitor. RBP elicited broadly t... |
|
|
Molecular mechanism of the inhibition of cytochrome c aggregation by Phe-Gly.
Arch. Biochem. Biophys. 435(1) , 182-9, (2005) Experimental and computational studies suggest that few general principles govern protein/protein interactions and aggregation. The knowledge of these rules may be exploited to design peptides that ar... |
|
|
Intrinsic folding of small peptide chains: spectroscopic evidence for the formation of beta-turns in the gas phase.
J. Am. Chem. Soc. 127(2) , 707-12, (2005) Laser desorption of model peptides coupled to laser spectroscopic techniques enables the gas-phase observation of genuine secondary structures of biology. Spectroscopic evidence for the formation of b... |
| EINECS 211-958-6 |
| N-Glycyl-DL-B-phenylalanine |
| Gly-D-Phe-OH/Gly-L-Phe-OH,(1:1) |
| Glycylphenylalanine |
| 2-(glycylamino)-3-phenyl-propionic acid |
| N-glycyl-DL-phenylalanine |
| N-glycyl-3-phenyl-DL-alanine |
| H-GLY-DL-PHE-OH |
| N-glycyl-phenylalanine |
| MFCD00065109 |
| GLY-DL-PHE |
| rac-(R*)-N-Glycyl-2-benzylglycine |
![3-phenyl-2-[(2-phenylmethoxycarbonylaminoacetyl)amino]propanoic acid structure](https://www.chemsrc.com/caspic/287/5540-03-4.png)

![Phenylalanine,N-[(1,3-dihydro-1,3-dioxo-2H-isoindol-2-yl)acetyl]- (9CI) structure](https://www.chemsrc.com/caspic/234/60676-54-2.png)