Fmoc-Lys(Boc)-OH
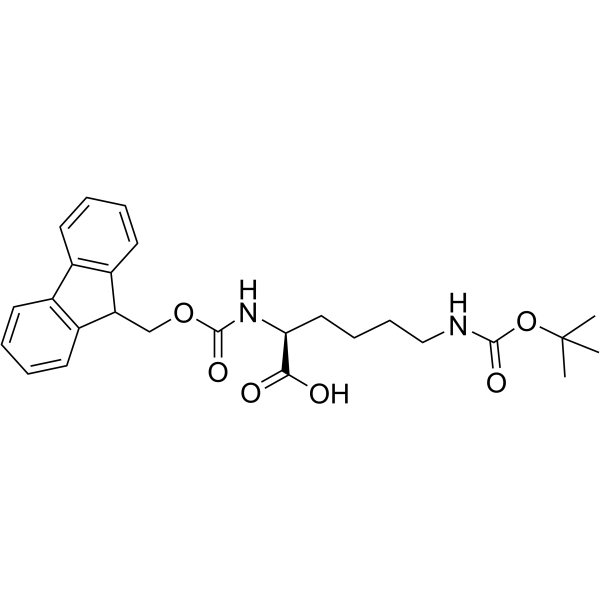
Fmoc-Lys(Boc)-OH structure
|
Common Name | Fmoc-Lys(Boc)-OH | ||
|---|---|---|---|---|
| CAS Number | 71989-26-9 | Molecular Weight | 468.542 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 685.7±55.0 °C at 760 mmHg | |
| Molecular Formula | C26H32N2O6 | Melting Point | 130-135 °C (dec.) | |
| MSDS | Chinese USA | Flash Point | 368.5±31.5 °C | |
Use of Fmoc-Lys(Boc)-OHFmoc-L-Lys (Boc)-OH is a lysine derivative[1]. |
| Name | (2S)-2-(9H-fluoren-9-ylmethoxycarbonylamino)-6-[(2-methylpropan-2-yl)oxycarbonylamino]hexanoic acid |
|---|---|
| Synonym | More Synonyms |
| Description | Fmoc-L-Lys (Boc)-OH is a lysine derivative[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Amino acids and amino acid derivatives have been commercially used as ergogenic supplements. They influence the secretion of anabolic hormones, supply of fuel during exercise, mental performance during stress related tasks and prevent exercise induced muscle damage. They are recognized to be beneficial as ergogenic dietary substances[1]. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 685.7±55.0 °C at 760 mmHg |
| Melting Point | 130-135 °C (dec.) |
| Molecular Formula | C26H32N2O6 |
| Molecular Weight | 468.542 |
| Flash Point | 368.5±31.5 °C |
| Exact Mass | 468.226044 |
| PSA | 113.96000 |
| LogP | 4.97 |
| Vapour Pressure | 0.0±2.2 mmHg at 25°C |
| Index of Refraction | 1.566 |
|
~% 
Fmoc-Lys(Boc)-OH CAS#:71989-26-9 |
| Literature: Chemical and Pharmaceutical Bulletin, , vol. 47, # 10 p. 1489 - 1490 |
|
~% 
Fmoc-Lys(Boc)-OH CAS#:71989-26-9 |
| Literature: Chemical and Pharmaceutical Bulletin, , vol. 47, # 10 p. 1489 - 1490 |
|
~% 
Fmoc-Lys(Boc)-OH CAS#:71989-26-9 |
| Literature: Chemical and Pharmaceutical Bulletin, , vol. 47, # 10 p. 1489 - 1490 |
| Precursor 4 | |
|---|---|
| DownStream 10 | |
| HS Code | 2924299090 |
|---|---|
| Summary | 2924299090. other cyclic amides (including cyclic carbamates) and their derivatives; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Quantification of plasma HIV RNA using chemically engineered peptide nucleic acids.
Nat. Commun. 5 , 5079, (2014) The remarkable stability of peptide nucleic acids (PNAs) towards enzymatic degradation makes this class of molecules ideal to develop as part of a diagnostic device. Here we report the development of ... |
|
|
Diels-Alder hydrogels with enhanced stability: First step toward controlled release of bevacizumab.
Eur. J. Pharm. Biopharm. 96 , 217-25, (2015) Eight-armed PEG was functionalized with furyl and maleimide groups (8armPEG20k-Fur and 8armPEG20k-Mal); degradable hydrogels were obtained by cross-linking via Diels-Alder chemistry. To increase the s... |
|
|
Chemical synthesis of a polypeptide backbone derived from the primary sequence of the cancer protein NY-ESO-1 enabled by kinetically controlled ligation and pseudoprolines.
Biopolymers 104 , 116-27, (2015) The cancer protein NY-ESO-1 has been shown to be one of the most promising vaccine candidates although little is known about its cellular function. Using a chemical protein strategy, the 180 amino aci... |
| Na-Fmoc-Ne-Boc-L-lysine |
| N|A-Fmoc-N|A-Boc-L-lysine |
| EINECS 276-256-4 |
| N-Fmoc-N'-Boc-L-Lysine |
| Nepsilon-Boc-Nalpha-Fmoc-L-lysine |
| L-Lysine, N-[(1,1-dimethylethoxy)carbonyl]-N-[(9H-fluoren-9-ylmethoxy)carbonyl]- |
| N-[(9H-Fluoren-9-ylmethoxy)carbonyl]-N-{[(2-methyl-2-propanyl)oxy]carbonyl}-L-lysine |
| N-α-FMOC-N-ε-tert-boc-L-lysine |
| Fmoc-Lys(Boc) |
| Fmoc-Lys(Boc)-OH |
| N6-(tert-Butoxycarbonyl)-N2-((9H-fluoren-9-ylmethoxy)carbonyl)-L-lysine |
| MFCD00037138 |
| (2S)-2-{[(9H-Fluoren-9-ylmethoxy)carbonyl]amino}-6-({[(2-methyl-2-propanyl)oxy]carbonyl}amino)hexanoic acid |
| N-alpha-FMOC-Nepsilon-BOC-L-Lysine |
| N-(tert-Butoxycarbonyl)-N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-lysine |
| N-(9-fluorenyl)methoxycarbonyl-Lys(Boc)-OH |
 CAS#:49557-75-7
CAS#:49557-75-7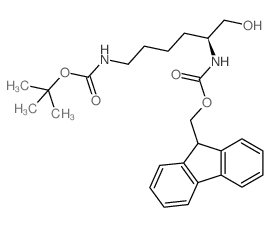 CAS#:198561-38-5
CAS#:198561-38-5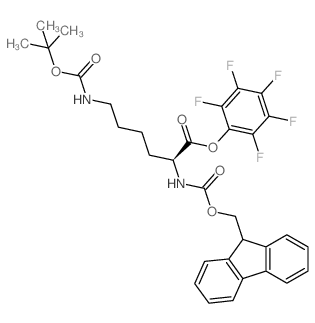 CAS#:86060-98-2
CAS#:86060-98-2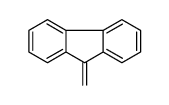 CAS#:4425-82-5
CAS#:4425-82-5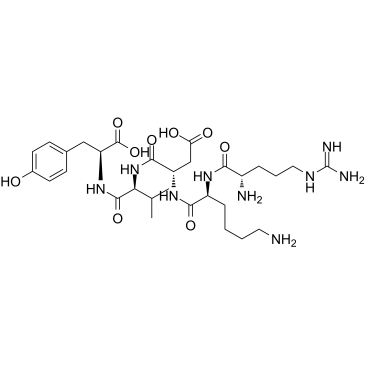 CAS#:69558-55-0
CAS#:69558-55-0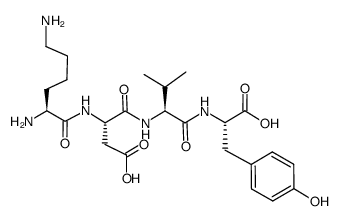 CAS#:75957-56-1
CAS#:75957-56-1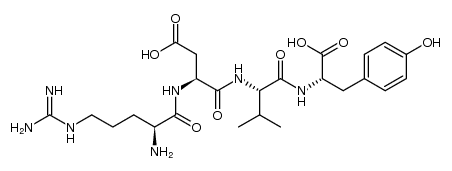 CAS#:1207730-31-1
CAS#:1207730-31-1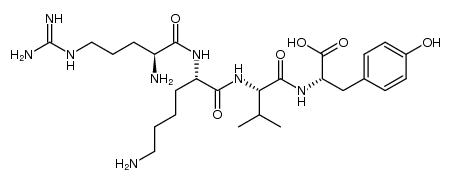 CAS#:1207730-30-0
CAS#:1207730-30-0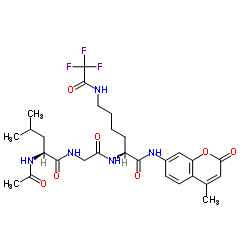 CAS#:1026295-98-6
CAS#:1026295-98-6
