ZLDI-8
Modify Date: 2024-01-17 17:24:13
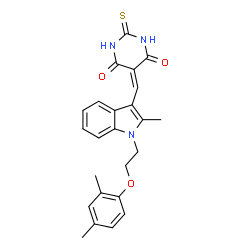
ZLDI-8 structure
|
Common Name | ZLDI-8 | ||
|---|---|---|---|---|
| CAS Number | 667880-38-8 | Molecular Weight | 433.523 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C24H23N3O3S | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
Use of ZLDI-8ZLDI-8 (IAC-8) is a novel Notch signaling pathway inhibitor for Notch activating/cleaving enzyme ADAM-17, significantly decreases the level of NICD and accumulation of NICD in the nucleus; exhibits cytotoxic acitviity against MHCC97-H cells with IC50 of 5.32 uM, reduces the expression of pro-survival/anti-apoptosis regulators, Survivin and cIAP1/2, also increases the expression of epithelial marker E-Cadherin and reduces mesenchymal markers N-Cadherin and Vimentin in HCC cells; significantly disrupted the activity of Notch pathway in HCC cells and inhibits the epithelial-mesenchymal transition (EMT) process of HCC cells; ZLDI-8 treatment enhances the susceptibility of HCC cells to Sorafenib, Etoposide, and Paclitaxel both in vitro and in vivo. |
| Name | 5-({1-[2-(2,4-Dimethylphenoxy)ethyl]-2-methyl-1H-indol-3-yl}methylene)-2-thioxodihydro-4,6(1H,5H)-pyrimidinedione |
|---|---|
| Synonym | More Synonyms |
| Description | ZLDI-8 (IAC-8) is a novel Notch signaling pathway inhibitor for Notch activating/cleaving enzyme ADAM-17, significantly decreases the level of NICD and accumulation of NICD in the nucleus; exhibits cytotoxic acitviity against MHCC97-H cells with IC50 of 5.32 uM, reduces the expression of pro-survival/anti-apoptosis regulators, Survivin and cIAP1/2, also increases the expression of epithelial marker E-Cadherin and reduces mesenchymal markers N-Cadherin and Vimentin in HCC cells; significantly disrupted the activity of Notch pathway in HCC cells and inhibits the epithelial-mesenchymal transition (EMT) process of HCC cells; ZLDI-8 treatment enhances the susceptibility of HCC cells to Sorafenib, Etoposide, and Paclitaxel both in vitro and in vivo. |
|---|---|
| Related Catalog | |
| Target |
ADAM-17[1] IC50: 31.6 μM (Tyrosine phosphatase)[1] Ki: 26.22 μM (Tyrosine phosphatase)[1] |
| In Vitro | ZLDI-8 (0.03-30 μM; 6-72 hours; MHCC97-H cells) treatment reduces cell viability in a time- and dose-dependent manner[1]. ZLDI-8 (1-10 μM; 6-72 hours; MHCC97-H cells) significantly decreases the level of NICD and the accumulation of NICD in the nucleus. ZLDI-8 could also reduce the expression of pro-survival/anti-apoptosis regulators, Survivin and cIAP1/2. And also increases the expression of epithelial marker E-Cadherin and reduced mesenchymal markers N-Cadherin and Vimentin[1]. ZLDI-8 enhances chemotherapy effects on tumor cell proliferation blockage, induction of apoptosis and cell-cycle arrest by inhibiting Notch pathway and blocking chemical resistance[1]. Cell Viability Assay[1] Cell Line: MHCC97-H cells Concentration: 0.03 μM, 0.1 μM, 0.3 μM, 1 μM, 3 μM, 10 μM, 30 μM Incubation Time: 6 hours, 12 hours, 24 hours, 48 hours, 72 hours Result: Emerged cytotoxic effect on MHCC97-H cells in a time- and dose-dependent manner. Western Blot Analysis[1] Cell Line: MHCC97-H cells Concentration: 1 μM, 3 μM, 10 μM Incubation Time: 6 hours, 12 hours, 24 hours, 48 hours, 72 hours Result: Significantly decreased the level of NICD and the accumulation of NICD in the nucleus. Also reduced the expression of pro-survival/anti-apoptosis regulators, Survivin and cIAP1/2 |
| In Vivo | ZLDI-8 (0.2-2 mg/kg; intraperitoneal injection; every two days; for 20 days; nude mice) treatment enhances the effect of Sorafenib on inhibiting tumor growth in nude HCC-bearing mice model[1]. Animal Model: Nude mice with MHCC-97H cells[1] Dosage: 2 mg/kg, 1 mg/kg, 500 μg/kg, or 200 μg/kg Administration: Intraperitoneal injection; every two days; for 20 days Result: Inhibited tumor growth in nude HCC-bearing mice model. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Molecular Formula | C24H23N3O3S |
| Molecular Weight | 433.523 |
| Exact Mass | 433.146027 |
| LogP | 5.26 |
| Index of Refraction | 1.662 |
| Hazard Codes | Xi |
|---|
| 4,6(1H,5H)-Pyrimidinedione, 5-[[1-[2-(2,4-dimethylphenoxy)ethyl]-2-methyl-1H-indol-3-yl]methylene]dihydro-2-thioxo- |
| 5-({1-[2-(2,4-Dimethylphenoxy)ethyl]-2-methyl-1H-indol-3-yl}methylene)-2-thioxodihydro-4,6(1H,5H)-pyrimidinedione |