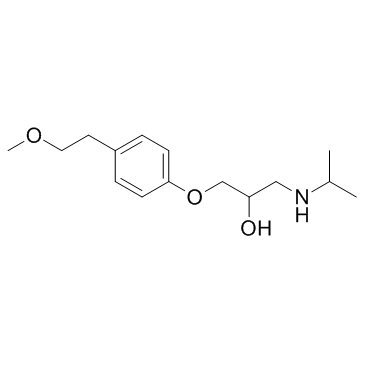
UNII:C19D0413EL
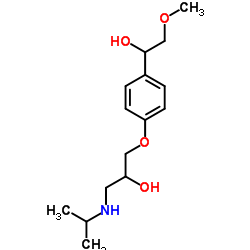
UNII:C19D0413EL structure
|
Common Name | UNII:C19D0413EL | ||
|---|---|---|---|---|
| CAS Number | 56392-16-6 | Molecular Weight | 283.363 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 437.6±45.0 °C at 760 mmHg | |
| Molecular Formula | C15H25NO4 | Melting Point | 65-67ºC | |
| MSDS | USA | Flash Point | 218.4±28.7 °C | |
Use of UNII:C19D0413ELa-Hydroxymetoprolol is a metabolite of metoprolol. The adrenoreceptor blocking effect of a-Hydroxymetoprolol on metoprolol is almost zero[1]. |
| Name | α-Hydroxy Metoprolol |
|---|---|
| Synonym | More Synonyms |
| Description | a-Hydroxymetoprolol is a metabolite of metoprolol. The adrenoreceptor blocking effect of a-Hydroxymetoprolol on metoprolol is almost zero[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 437.6±45.0 °C at 760 mmHg |
| Melting Point | 65-67ºC |
| Molecular Formula | C15H25NO4 |
| Molecular Weight | 283.363 |
| Flash Point | 218.4±28.7 °C |
| Exact Mass | 283.178345 |
| PSA | 70.95000 |
| LogP | 0.72 |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.524 |
| Storage condition | -20°C Freezer |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| RIDADR | NONH for all modes of transport |
|---|
|
~95% 
UNII:C19D0413EL CAS#:56392-16-6 |
| Literature: Shetty, H. Umesha; Nelson, Wendel L. Journal of Medicinal Chemistry, 1988 , vol. 31, # 1 p. 55 - 59 |
|
~% 
UNII:C19D0413EL CAS#:56392-16-6 |
| Literature: Journal of Medicinal Chemistry, , vol. 31, # 1 p. 55 - 59 |
|
~% 
UNII:C19D0413EL CAS#:56392-16-6 |
| Literature: Journal of Medicinal Chemistry, , vol. 31, # 1 p. 55 - 59 |
|
~% 
UNII:C19D0413EL CAS#:56392-16-6 |
| Literature: Journal of Medicinal Chemistry, , vol. 31, # 1 p. 55 - 59 |
|
~% 
UNII:C19D0413EL CAS#:56392-16-6 |
| Literature: Journal of Medicinal Chemistry, , vol. 31, # 1 p. 55 - 59 |
|
~% 
UNII:C19D0413EL CAS#:56392-16-6 |
| Literature: Journal of Medicinal Chemistry, , vol. 31, # 1 p. 55 - 59 |
|
~% 
UNII:C19D0413EL CAS#:56392-16-6 |
| Literature: European Journal of Clinical Pharmacology, , vol. 59, # 5-6 p. 429 - 442 |
|
Simultaneous determination of metoprolol and α-hydroxymetoprolol in human plasma using excitation-emission matrix fluorescence coupled with second-order calibration methods.
Bioanalysis 4(23) , 2781-93, (2012) Metoprolol (MET) is a β1-adrenoceptor antagonist, which is widely used in the treatment of cardiovascular diseases, and α-hydroxymetoprolol (α-OHM) is its hydroxylated metabolite. Owing to their simil... |
|
|
Metoprolol oxidation polymorphism in Brazilian elderly cardiac patients.
J. Pharm. Pharmacol. 65(9) , 1347-53, (2013) The purpose of this study was to phenotype the CYP2D6 in elderly with heart disease classified as extensive metabolizer or poor metabolizers (PM) of metoprolol, develop and validate the method of anal... |
|
|
Variation of pharmacokinetics after oral administration of slow-release metoprolol tablets and pharmacogenetic considerations.
Arzneimittelforschung 42(6) , 802-6, (1992) The maximum plasma concentrations (Cmax) after oral administration of 120 mg tablets of slow-release metoprolol (CAS 37350-58-6) to 75 Japanese healthy male volunteers and 15 arrhythmic patients were ... |
| 1-[4-(1-Hydroxy-2-methoxyethyl)phenoxy]-3-(isopropylamino)-2-propanol |
| 3-[4-(1-Hydroxy-2-methoxyethyl)phenoxy]-1-isopropylamino-2-propanol |
| Benzenemethanol, 4-[2-hydroxy-3-[(1-methylethyl)amino]propoxy]-α-(methoxymethyl)- |
| α-HYDROXYMETOPROLOL |
| 1-[4-(1-hydroxy-2-methoxyethyl)phenoxy]-3-(isopropylamino)propan-2-ol |
| UNII:C19D0413EL |
![3-[4-(methylacetyl)phenoxy]-1-(isopropylamino)-2-propanol structure](https://www.chemsrc.com/caspic/120/110458-45-2.png)
![2-methoxy-1-[4-(oxiranylmethoxy)phenyl]ethanone structure](https://www.chemsrc.com/caspic/158/110458-44-1.png)
![3-[4-(methylacetyl)phenoxy]-1,2-propanediol structure](https://www.chemsrc.com/caspic/196/110458-43-0.png)