2,5-Furandicarboxylic acid

2,5-Furandicarboxylic acid structure
|
Common Name | 2,5-Furandicarboxylic acid | ||
|---|---|---|---|---|
| CAS Number | 3238-40-2 | Molecular Weight | 156.093 | |
| Density | 1.6±0.1 g/cm3 | Boiling Point | 419.2±30.0 °C at 760 mmHg | |
| Molecular Formula | C6H4O5 | Melting Point | >310°C (dec.) | |
| MSDS | Chinese USA | Flash Point | 207.3±24.6 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of 2,5-Furandicarboxylic acid2,5-Furandicarboxylic acid is a biomass-derived diacid that can be used to make polymers including polyethylene furandicarboxylate (PEF). |
| Name | furan-2,5-dicarboxylic acid |
|---|---|
| Synonym | More Synonyms |
| Description | 2,5-Furandicarboxylic acid is a biomass-derived diacid that can be used to make polymers including polyethylene furandicarboxylate (PEF). |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite |
| References |
| Density | 1.6±0.1 g/cm3 |
|---|---|
| Boiling Point | 419.2±30.0 °C at 760 mmHg |
| Melting Point | >310°C (dec.) |
| Molecular Formula | C6H4O5 |
| Molecular Weight | 156.093 |
| Flash Point | 207.3±24.6 °C |
| Exact Mass | 156.005875 |
| PSA | 87.74000 |
| LogP | -0.02 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.581 |
| Storage condition | Hygroscopic, -20°C Freezer, Under Inert Atmosphere |
| Stability | Light Sensitive, Very Hygroscopic |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H319 |
| Precautionary Statements | P280-P305 + P351 + P338-P337 + P313 |
| Hazard Codes | Xi |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S36/37/39 |
| RIDADR | NONH for all modes of transport |
| HS Code | 2932190090 |
| Precursor 10 | |
|---|---|
| DownStream 9 | |
| HS Code | 2932190090 |
|---|---|
| Summary | 2932190090 other compounds containing an unfused furan ring (whether or not hydrogenated) in the structure VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:6.5% General tariff:20.0% |
|
Age-related reference values for urinary organic acids in a healthy Turkish pediatric population.
Clin. Chem. 40(6) , 862-6, (1994) Organic acid concentrations were quantified by gas chromatography and the individual acids identified by mass spectrometry in urine specimens from a healthy Turkish pediatric population of ages 2 days... |
|
|
Carbon dioxide utilization via carbonate-promoted C-H carboxylation.
Nature 531 , 215-9, (2016) Using carbon dioxide (CO2) as a feedstock for commodity synthesis is an attractive means of reducing greenhouse gas emissions and a possible stepping-stone towards renewable synthetic fuels. A major i... |
|
|
The identification and metabolic origin of 2-furoylglycine and 2,5-furandicarboxylic acid in human urine.
Clin. Chim. Acta 41 , 199-207, (1972)
|
| Furan 2,5-dicarboxylic acid |
| Dehydroschleimsaeure |
| Dehydromucate |
| MFCD00016582 |
| Dehydromucic acid |
| 2,5-furandicarboxylate |
| 2,5-dicarboxyfuran |
| furan-2,5-dicarbonsaeure |
| Furan-2,5-dicarboxylic acid |
| EINECS 221-800-8 |
| 2,5-Furandicarboxylic acid |
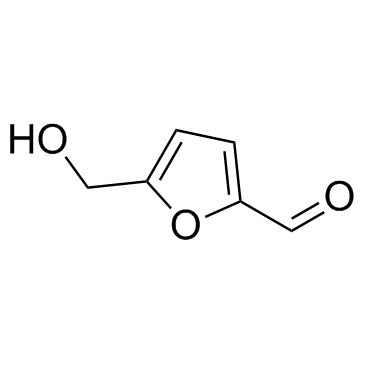 CAS#:67-47-0
CAS#:67-47-0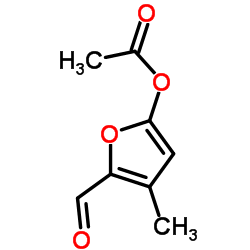 CAS#:10551-58-3
CAS#:10551-58-3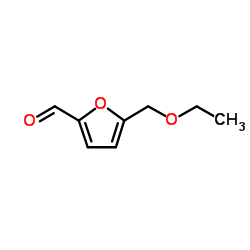 CAS#:1917-65-3
CAS#:1917-65-3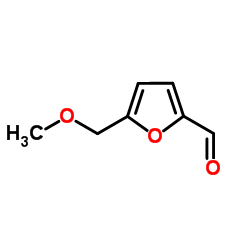 CAS#:1917-64-2
CAS#:1917-64-2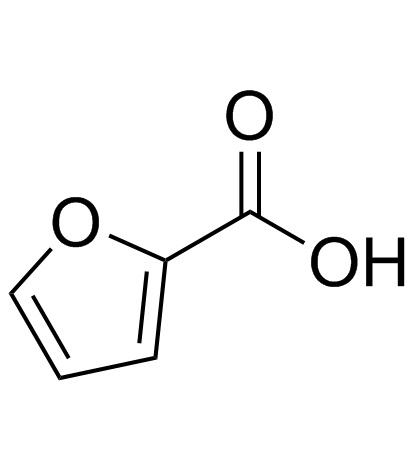 CAS#:88-14-2
CAS#:88-14-2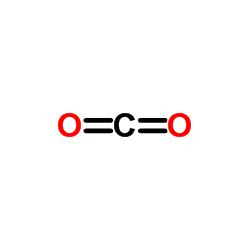 CAS#:124-38-9
CAS#:124-38-9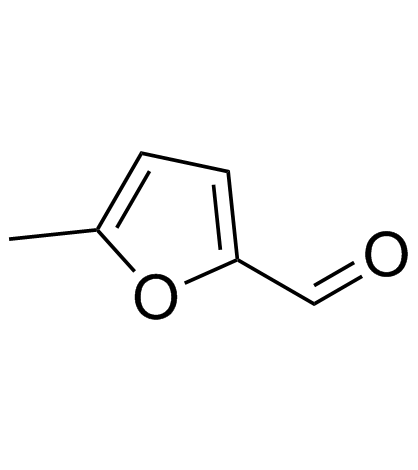 CAS#:620-02-0
CAS#:620-02-0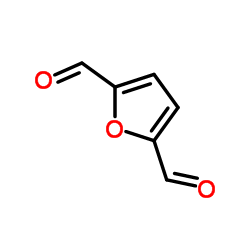 CAS#:823-82-5
CAS#:823-82-5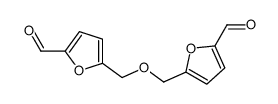 CAS#:7389-38-0
CAS#:7389-38-0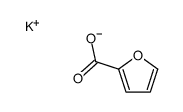 CAS#:20842-02-8
CAS#:20842-02-8 CAS#:56286-72-7
CAS#:56286-72-7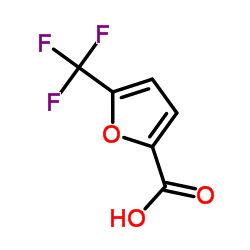 CAS#:56286-73-8
CAS#:56286-73-8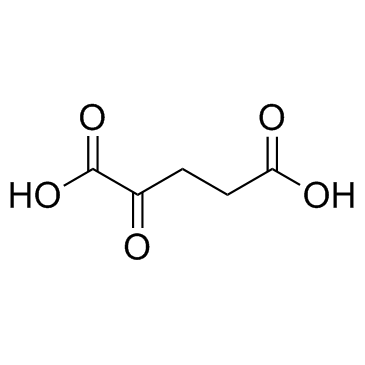 CAS#:328-50-7
CAS#:328-50-7 CAS#:110-15-6
CAS#:110-15-6 CAS#:4282-32-0
CAS#:4282-32-0 CAS#:110-17-8
CAS#:110-17-8 CAS#:53662-83-2
CAS#:53662-83-2 CAS#:645-12-5
CAS#:645-12-5
