H-Gly-OtBu·HCl
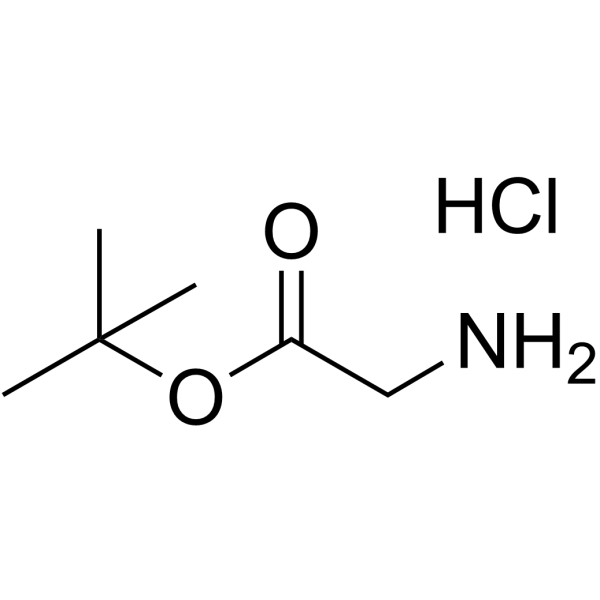
H-Gly-OtBu·HCl structure
|
Common Name | H-Gly-OtBu·HCl | ||
|---|---|---|---|---|
| CAS Number | 27532-96-3 | Molecular Weight | 167.634 | |
| Density | 0.973g/cm3 | Boiling Point | 145.7ºC at 760mmHg | |
| Molecular Formula | C6H14ClNO2 | Melting Point | 141-143 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 20.7ºC | |
Use of H-Gly-OtBu·HClGlycine tert-butyl ester hydrochloride is a Glycine (HY-Y0966) derivative[1]. |
| Name | tert-butyl 2-aminoacetate,hydrochloride |
|---|---|
| Synonym | More Synonyms |
| Description | Glycine tert-butyl ester hydrochloride is a Glycine (HY-Y0966) derivative[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Amino acids and amino acid derivatives have been commercially used as ergogenic supplements. They influence the secretion of anabolic hormones, supply of fuel during exercise, mental performance during stress related tasks and prevent exercise induced muscle damage. They are recognized to be beneficial as ergogenic dietary substances[1]. |
| References |
| Density | 0.973g/cm3 |
|---|---|
| Boiling Point | 145.7ºC at 760mmHg |
| Melting Point | 141-143 °C(lit.) |
| Molecular Formula | C6H14ClNO2 |
| Molecular Weight | 167.634 |
| Flash Point | 20.7ºC |
| Exact Mass | 167.071304 |
| PSA | 52.32000 |
| LogP | 1.78910 |
| Vapour Pressure | 0.026mmHg at 25°C |
| Index of Refraction | 1.522 |
| Storage condition | −20°C |
| Water Solubility | H2O: 0.1 g/mL, clear, colorless |
| Precursor 0 | |
|---|---|
| DownStream 10 | |
| HS Code | 2922499990 |
|---|---|
| Summary | HS:2922499990 other amino-acids, other than those containing more than one kind of oxygen function, and their esters; salts thereof VAT:17.0% Tax rebate rate:9.0% Supervision conditions:AB(certificate of inspection for goods inward,certificate of inspection for goods outward) MFN tariff:6.5% General tariff:30.0% |
|
Inhibition of angiogenesis by antioxidant micelles.
Adv. Healthc. Mater. 4(4) , 569-75, (2015) Antioxidant micelles capable of scavenging reactive oxygen species (ROS) are prepared from poly(ethylene glycol)-b-poly(dopamine) block copolymers. The micelles inhibit tube formation of human umbilic... |
|
|
Synthesis and characterization of more potent analogues of hirudin fragment 1-47 containing non-natural amino acids.
Biochemistry 37(39) , 13507-15, (1998) Hirudin is the most potent and specific inhibitor of thrombin, a key enzyme in the coagulation process existing in equilibrium between its procoagulant (fast) and anticoagulant (slow) form. In a previ... |
|
|
Chiral phosphine-free Pd-mediated asymmetric allylation of prochiral enolate with a chiral phase-transfer catalyst.
Org. Lett. 3(21) , 3329-31, (2001) [reaction: see text]. A chiral phase-transfer catalyst has been applied to the asymmetric allylation of the tert-butyl glycinate-benzophenone Schiff base with various allylic acetates for the first ti... |
| 2-tert-Butoxy-2-oxoethanaminium chloride |
| H-Gly-OtBu.HCl |
| Glycine tert-butyl ester hydrochloride |
| Glycine tertbutyl ester hydrochloride |
| tert-butylglycine ester |
| tert-Butyl glycinate hydrochloride (1:1) |
| H-Gly-OtBu,HCl |
| tert-Butyl aminoacetate hydrochloride |
| Glycine, 1,1-dimethylethyl ester, hydrochloride (1:1) |
| t-butyl aminoacetate hydrochloride |
| hydrochoride |
| glycine 1,1-dimethylethyl ester hydrochloride |
| 2-Methyl-2-propanyl aminoacetate hydrochloride |
| tert-butyl glycinate hydrochloride |
| tert-Butyl 2-aminoacetate hydrochloride |
| EINECS 214-223-8 |
| tert-butyl glycine hydrochloride |
| H-Gly-OtBu·HCl |
| tert-butyl 2-aminoacetate,chloride |
| H2N-Gly-OtBu*HCl |
| Glycine t-butyl ester hydrochloride |
| 2-Methyl-2-propanyl glycinate hydrochloride (1:1) |
| t-Butyl glycinate |
| MFCD00058255 |
| 2-Methyl-2-propanyl glycinate hydrochloride |
| Glycine tert butyl ester hydrochloride |
| GLYCINE TERT-BUTYL ESTER HCl |
![2-Methyl-2-propanyl ({[(2-methyl-2-propanyl)oxy]carbonyl}amino)(p henyl)acetate structure](https://www.chemsrc.com/caspic/454/111652-10-9.png) CAS#:111652-10-9
CAS#:111652-10-9 CAS#:35059-50-8
CAS#:35059-50-8 CAS#:81477-94-3
CAS#:81477-94-3 CAS#:6456-74-2
CAS#:6456-74-2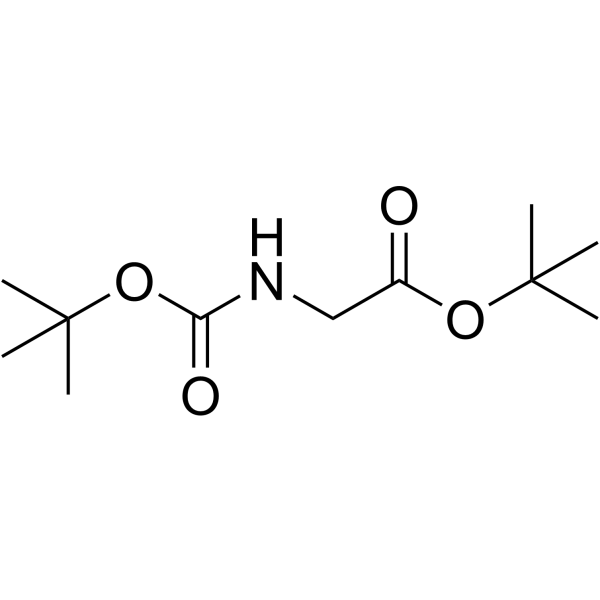 CAS#:111652-20-1
CAS#:111652-20-1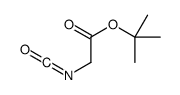 CAS#:113238-61-2
CAS#:113238-61-2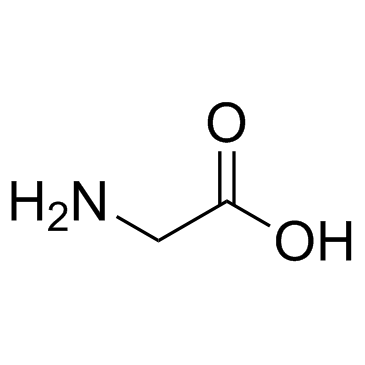 CAS#:56-40-6
CAS#:56-40-6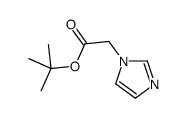 CAS#:83468-75-1
CAS#:83468-75-1 CAS#:84117-86-2
CAS#:84117-86-2![tert-butyl N-((2R)-2-{[(benzyloxy)carbonyl]amino}-2-phenylethanoyl)glycinate structure](https://www.chemsrc.com/caspic/294/439088-73-0.png) CAS#:439088-73-0
CAS#:439088-73-0
