5-Fluorouracil-13C4,15N2
Modify Date: 2024-04-03 18:47:35
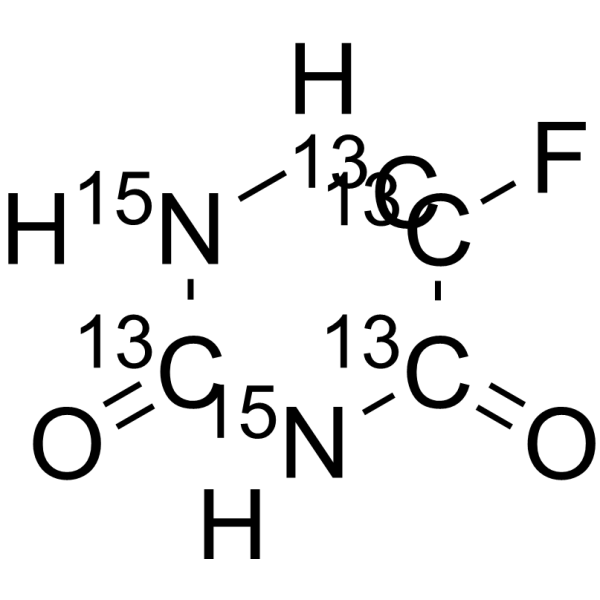
5-Fluorouracil-13C4,15N2 structure
|
Common Name | 5-Fluorouracil-13C4,15N2 | ||
|---|---|---|---|---|
| CAS Number | 202407-03-2 | Molecular Weight | 136.03 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | 13C4H3F15N2O2 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
Use of 5-Fluorouracil-13C4,15N25-Fluorouracil-13C4,15N2 is the 13C and 15N labeled 5-Fluorouracil[1]. 5-Fluorouracil (5-FU) is an analogue of uracil and a potent antitumor agent. 5-Fluorouracil affects pyrimidine synthesis by inhibiting thymidylate synthetase thus depleting intracellular dTTP pools. 5-Fluorouracil induces apoptosis and can be used as a chemical sensitizer[2][3]. 5-Fluorouracil also inhibits HIV[4]. |
| Name | 5-Fluorouracil-13C4,15N2 |
|---|
| Description | 5-Fluorouracil-13C4,15N2 is the 13C and 15N labeled 5-Fluorouracil[1]. 5-Fluorouracil (5-FU) is an analogue of uracil and a potent antitumor agent. 5-Fluorouracil affects pyrimidine synthesis by inhibiting thymidylate synthetase thus depleting intracellular dTTP pools. 5-Fluorouracil induces apoptosis and can be used as a chemical sensitizer[2][3]. 5-Fluorouracil also inhibits HIV[4]. |
|---|---|
| Related Catalog | |
| In Vitro | Stable heavy isotopes of hydrogen, carbon, and other elements have been incorporated into drug molecules, largely as tracers for quantitation during the drug development process. Deuteration has gained attention because of its potential to affect the pharmacokinetic and metabolic profiles of drugs[1]. |
| References |
| Molecular Formula | 13C4H3F15N2O2 |
|---|---|
| Molecular Weight | 136.03 |