trans-Glutaconic acid
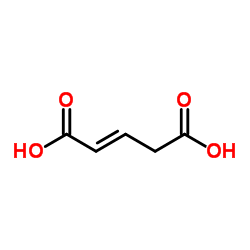
trans-Glutaconic acid structure
|
Common Name | trans-Glutaconic acid | ||
|---|---|---|---|---|
| CAS Number | 1724-02-3 | Molecular Weight | 130.099 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 413.8±28.0 °C at 760 mmHg | |
| Molecular Formula | C5H6O4 | Melting Point | 133-135ºC | |
| MSDS | Chinese USA | Flash Point | 218.2±20.5 °C | |
| Name | glutaconic acid |
|---|---|
| Synonym | More Synonyms |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 413.8±28.0 °C at 760 mmHg |
| Melting Point | 133-135ºC |
| Molecular Formula | C5H6O4 |
| Molecular Weight | 130.099 |
| Flash Point | 218.2±20.5 °C |
| Exact Mass | 130.026611 |
| PSA | 74.60000 |
| LogP | 0.04 |
| Vapour Pressure | 0.0±2.1 mmHg at 25°C |
| Index of Refraction | 1.517 |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Safety Phrases | S22-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2917190090 |
| Precursor 10 | |
|---|---|
| DownStream 9 | |
| HS Code | 2917190090 |
|---|---|
| Summary | 2917190090 acyclic polycarboxylic acids, their anhydrides, halides, peroxides, peroxyacids and their derivatives VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:6.5% General tariff:30.0% |
|
Stereodifferentiation of 3-hydroxyisobutyric- and 3-aminoisobutyric acid in human urine by enantioselective multidimensional capillary gas chromatography-mass spectrometry.
Clin. Chim. Acta 292 , 93-105, (2000) The chiral metabolites 3-hydroxyisobutyric acid (HIBA) and 3-aminoisobutyric acid (AIBA) are intermediates in the pathways of L-valine and thymine and play an important role in the diagnosis of the ve... |
|
|
Induction of the mitochondrial permeability transition in vitro by short-chain carboxylic acids.
Biochem. Biophys. Res. Commun. 272(2) , 431-5, (2000) We recently reported that acrylic acid (AA) induces the MPT in vitro, which we suggested might be a critical event in the acute inflammatory and hyperplastic response of the olfactory epithelium. The ... |
|
|
Efficient synthesis and neuroprotective effect of substituted 1,3-diphenyl-2-propen-1-ones.
J. Med. Chem. 51 , 4054-8, (2008) An efficient synthesis involving a key aldol reaction and biological properties of 1,3-diphenyl-2-propen-1-ones 8- 20 is described. The in vitro activity for 2,2-diphenyl-1-picrylhydrazyl (DPPH) radic... |
| pentenedioic acid |
| EINECS 217-027-0 |
| Pentendisaeure |
| Glutaconic acid |
| pentene diacid |
| Pent-2-ene-1,5-dioic acid |
| TRANS-GLUTACONIC ACID |
| (2E)-Pent-2-enedioic acid |
| (E)-glutaconic acid |
| (2E)-2-Pentenedioic acid |
| 2-Pentenedioic acid, (2E)- |
| pent-2-enedioic acid |
| 2-Pentenedioic acid |
| prop-1-ene-1,3-dicarboxylic acid |
| acide glutaconique |
| MFCD00002784 |
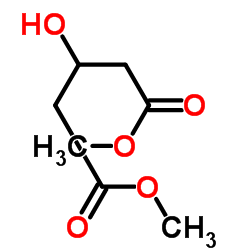 CAS#:7250-55-7
CAS#:7250-55-7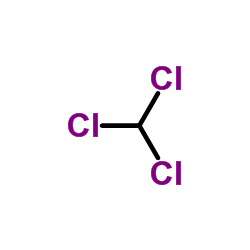 CAS#:67-66-3
CAS#:67-66-3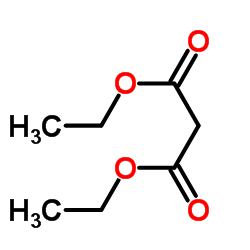 CAS#:105-53-3
CAS#:105-53-3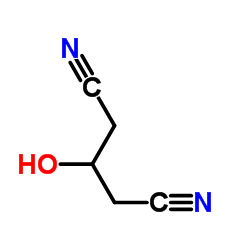 CAS#:13880-89-2
CAS#:13880-89-2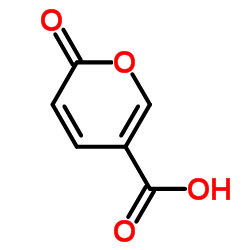 CAS#:500-05-0
CAS#:500-05-0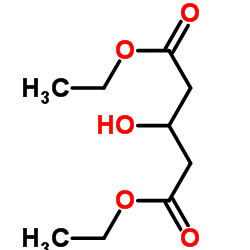 CAS#:32328-03-3
CAS#:32328-03-3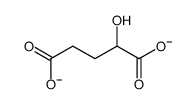 CAS#:2889-31-8
CAS#:2889-31-8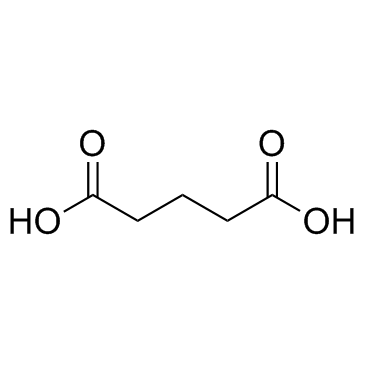 CAS#:110-94-1
CAS#:110-94-1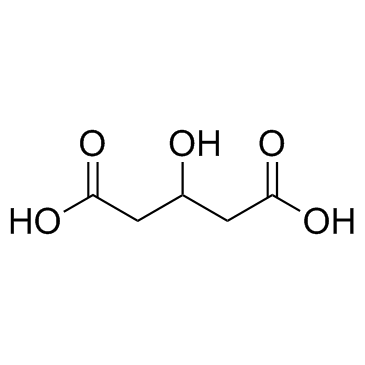 CAS#:638-18-6
CAS#:638-18-6 CAS#:7647-01-0
CAS#:7647-01-0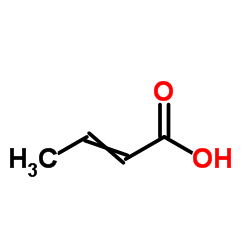 CAS#:3724-65-0
CAS#:3724-65-0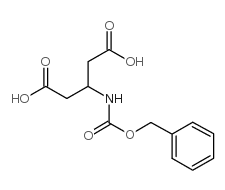 CAS#:17336-01-5
CAS#:17336-01-5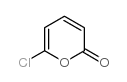 CAS#:20357-65-7
CAS#:20357-65-7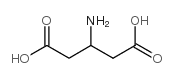 CAS#:1948-48-7
CAS#:1948-48-7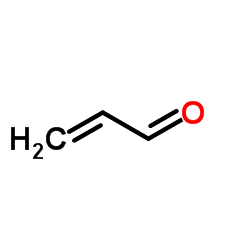 CAS#:107-02-8
CAS#:107-02-8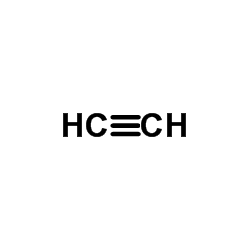 CAS#:74-86-2
CAS#:74-86-2 CAS#:4636-39-9
CAS#:4636-39-9